Read about the latest developments with epinephrine nasal sprays. Plus, check out our mythbuster on oral allergy/pollen food allergy syndrome.
Research: More developments with epinephrine nasal sprays
ARS Pharmaceuticals announced that the U.S. Food and Drug Administration (FDA) has scheduled an advisory committee meeting for May 11, 2023 to review the new drug application (NDA) for neffy®, the company’s epinephrine nasal spray. With an NDA, a pharmaceutical company makes a formal request for approval of a new medication. Read the press release for more details.

Bryn Pharma announced positive results from a study which assessed the impact of nasal congestion on the absorption of its UTULY™ epinephrine nasal spray. Results showed UTULY is absorbed as fast as with an auto-injector, and offers a higher and more sustained level of epinephrine. Read the press release for more details.
We’ll keep you updated as the news on needle-free epinephrine alternatives continue to evolve.
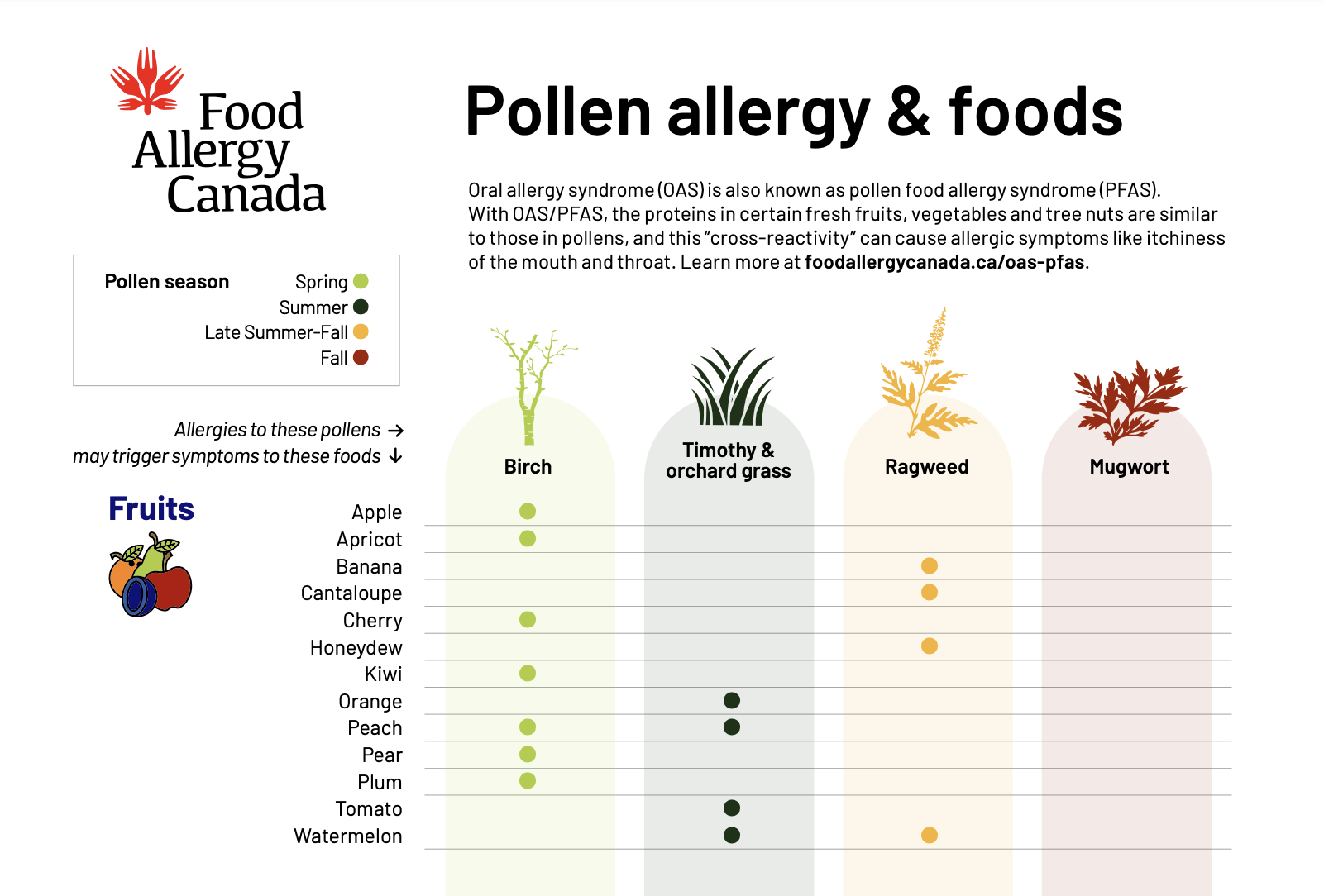
Mythbuster: Do oral allergy syndrome (OAS)/pollen food allergy syndrome (PFAS) symptoms only happen during pollen season?
FACT: No, not necessarily. With OAS/PFAS, the proteins in certain fresh fruits, vegetables and tree nuts are similar to those in pollens, and this “cross-reactivity” can cause allergic symptoms like itchiness of the mouth and throat. Symptoms of OAS/PFAS can happen all year long, although sometimes people notice more severe symptoms during pollen season.
Learn more
Help us educate your communities and share this mythbuster with them! Find more mythbusters at foodallergycanada.ca/mythbusters.
Tags: mythbuster, needle-free epinephrine, OAS, PFAS, Research